Research Organogram
Proposal submission instructions
Dear Researcher,
To submit your research protocol for ethical review, visit https://redcap15.moh.gov.zm/redcap/surveys/?s=AHHNMKKAWPPYXJPR. Protocols may be submitted in any appropriate format.
To facilitate timely processing and clearance by the LCPTS-REC, please ensure that all items on the submission link are uploaded. You will get an email confirmation from the secretariate within 24hrs.
Admin Review Fees
| Level/type of submission | Regular submission – Feedback within 1-2 weeks (ZMW) | Expedited – Feedback within 24-72hrs (ZMW) |
| Diploma/Certificate | 150 | 250 |
| Undergraduate Degree | 200 | 400 |
| Master’s Level | 500 | 1000 |
| PhD Level | 1000 | 2000 |
| Independent Non-Funded Researchers | 500 | 1000 |
| NGO / Funded Projects (non-clinical trials) | 2000 | 4000 |
| Clinical Trials | 4000 |
All payments should be made through the LCPTS institutional account below:
Account number: 0102030000641
Account Name: LIVINGSTONE CENTER FOR PREVENTION
Full Account name: LIVINGSTONE CENTER FOR PREVENTION AND TRANSLATIONAL SCIENCE
Indo Zambia Bank– LIVINGSTONE BRANCH
Sort Code: 091010
Swift Code: INZAZMLX
Currency: ZMW
Please attach a copy of the receipt as proof of payment. Reference on payment should indicate “Ethics review contribution”
For any clarification or guidance, contact the REC Secretariat via rec@lcpts.org or call +260 767 8181 99
Standard Operating Procedures (SOP)
LCPTS Research Ethics Committee (LCPTS-REC)
IORG0012647, FWA00035852; IRB00014955
Contact: rec@lcpts.org
Introduction
The LCPTS Research Ethics Committee (LCPTS-REC) ensures that any research involving human participants or animal subjects is conducted according to internationally recognized ethical principles and regulatory standards. This includes adherence to foundational documents and guidelines such as the Nuremberg Code (1947), the World Medical Association’s Declaration of Helsinki (1964, with subsequent revisions through 2013), the Council for International Organizations of Medical Sciences (CIOMS) International Ethical Guidelines (latest edition), the World Health Organization (WHO) and International Conference on Harmonisation (ICH) Good Clinical Practice (GCP) guidelines, and the Belmont Report (1979). The committee’s standards are also in line with U.S. federal regulations for human research protection (e.g., 45 CFR 46, “Common Rule”) and National Institutes of Health (NIH) policies on the responsible conduct of research, especially when U.S.-funded studies are involved. The LCPTS-REC likewise abides by national requirements, including Zambia’s National Health Research Act (2013) and any guidance issued by the National Health Research Authority (NHRA). Importantly, these SOPs encompass the ethical review of studies involving human participants and/or animals, in accordance with the Institutional Animal Care and Use Committee (IACUC) guidelines for animal research ethics.
Definition of Research: For the purposes of this SOP, research is defined as any systematic investigation—including development, testing, and evaluation—designed to develop or contribute to generalizable knowledge. This definition encompasses both:
- Therapeutic procedures: Interventions administered with the intent of providing direct benefit to the participant (for example, a clinical treatment expected to improve a patient’s condition as part of a trial).
- Non-therapeutic procedures: Interventions or interactions that are not intended to provide a direct health benefit to the participant, but rather are undertaken to answer a research question or test a hypothesis (for example, drawing an extra blood sample purely for laboratory analysis in a study).
Any activity that meets this definition is considered research under these guidelines, regardless of whether it is labeled or funded as research in other contexts. (For example, certain demonstration projects or service programs may include components that constitute research.) All health research conducted in Zambia is expected to comply with these ethical standards and be reviewed by an authorized Research Ethics Committee.
Objectives of the REC
The objective of the LCPTS-REC Standard Operating Procedures is to ensure that the committee functions effectively and consistently in its ethical review of health research proposals. By following these SOPs, the REC implements a thorough, uniform review process for all submitted research protocols, thereby safeguarding the rights, welfare, and well-being of research participants (human or animal). Ultimately, these procedures ensure that research conducted under LCPTS’s auspices is ethically sound and compliant with both local and international standards.
Role of the REC
The REC’s primary role is to protect the health, dignity, rights, and welfare of all human participants (and animals, where applicable) involved in research. This means the committee reviews every research proposal to ensure it adheres to established ethical principles, both international and local, such as respect for persons (autonomy), beneficence, and justice. The REC ensures that the pursuit of scientific knowledge never overtakes the safety and well-being of research participants.
In addition to protocol review, the LCPTS-REC provides ongoing ethical oversight for approved projects. It offers guidance and advice on research ethics to investigators, institutional leadership, policymakers, and other stakeholders as needed. The REC may also conduct or participate in community outreach and education efforts to raise awareness about ethical considerations in health research.
The LCPTS-REC operates under the oversight of Zambia’s national research ethics governance. It is registered with and guided by the National Health Research Ethics Board (NHREB) as mandated by the National Health Research Authority. In practice, this means that all studies must receive REC approval before commencing, and the REC submits reports of its activities to national authorities as required. Researchers are not permitted to start their studies at LCPTS or its partner institutions until they have obtained written ethical approval from the LCPTS-REC.
System of Ethical Review
National Framework: All institutional ethics committees in Zambia (including the LCPTS-REC) function within the national framework overseen by the National Health Research Ethics Board (NHREB) under the NHRA. The NHREB, appointed by the Ministry of Health, establishes policies and guidance for research ethics and monitors the conduct of research nationwide.
Local and National Jurisdiction: The NHREB specifies which types of research protocols require review at the national level and which can be reviewed by local (institutional) RECs. Typically, multi-center clinical trials or high-risk studies may need national-level review or clearance, while other studies can be handled by the local REC. The LCPTS-REC adheres to these guidelines. On occasion, the NHREB may request the LCPTS-REC to review a proposal on its behalf or provide a secondary opinion on a locally reviewed study. In all cases, the LCPTS-REC works in coordination with national authorities to ensure compliance with the country’s ethical standards.
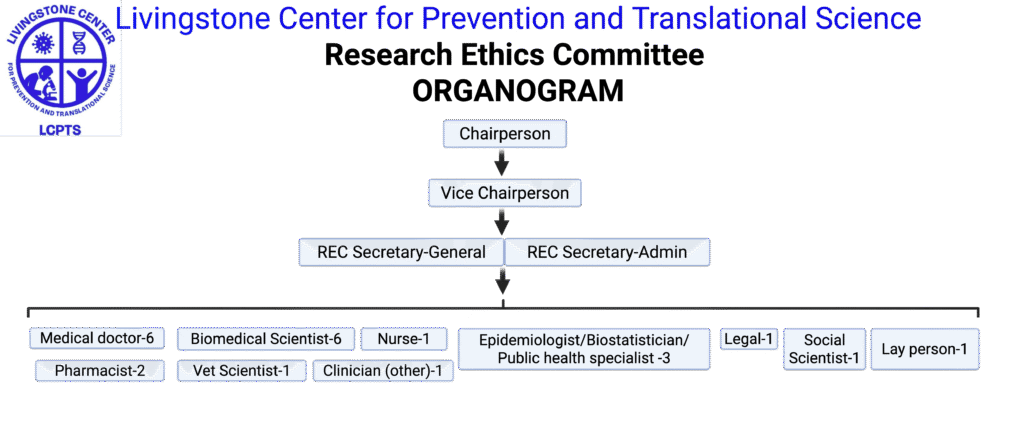
Composition
Diversity and Expertise: The LCPTS-REC is composed of members from diverse backgrounds to ensure a multi-disciplinary and multi-sectoral review perspective. The committee includes individuals with scientific expertise, clinicians, and members knowledgeable in ethics and law, as well as community representatives. Diversity in gender, age, and professional background is sought to reflect societal values and protect community interests.
Membership Roles: Members are appointed based on their personal expertise and integrity, not as delegates of their employers. At minimum, the committee’s composition includes:
- Chairperson: Leads the REC and facilitates meetings.
- Vice Chairperson: Assists the Chair and leads in the Chair’s absence.
- Secretary: Organizes meetings, prepares agendas and minutes, and manages documentation and communications.
- Vice Secretary: Assists the Secretary and can fulfill those duties if the Secretary is absent.
- Legal Expert: Provides insight on legal and regulatory issues in research.
- Religious/Community Representative: Offers perspective on community norms, cultural values, or religious considerations.
- Nursing or clinical officer/licentiate Professional: Brings practical healthcare experience and participant care perspective.
- Medical Scientist(s): Experts in biomedical or behavioral research.
- Clinician(s): Practicing medical doctor(s) and/or veterinarian(s) (to review studies involving human or animal health, respectively).
- Social Scientist/Biostatistician/Epidemiologist: Provides expertise in study design, statistics, or understanding of social contexts.
- Public Health/Ministry of Health Representative: Links the REC with public health priorities and regulatory expectations.
- Ethicist/Philosopher: Specialist in ethics or moral philosophy to provide guidance on ethical dilemmas.
- Layperson (Unaffiliated Community Member): An individual from the community with no scientific or institutional affiliation, to represent the perspective of the general public.
This diverse composition helps ensure a balance of scientific expertise and community input, safeguarding participant interests and public trust in the research process.
Membership Requirements
Terms of Service: Appointments to the LCPTS-REC are normally for a term of three years. To maintain continuity and institutional memory, terms are often renewable; however, to encourage fresh perspectives, the committee may stagger membership renewal such that approximately half of the members are replaced or reappointed every three years. Members should ideally not serve more than two consecutive terms (six years) without a break, unless their continued service is deemed essential.
Qualifications and Training: All REC members must have relevant qualifications or experience that enable them to review research protocols. Members are generally expected to have documented training in research ethics. The NHRA or other recognized bodies may provide certification or accreditation for REC members. LCPTS-REC members might be required, for example, to complete an ethics training course (such as the CITI program or equivalent) as part of their eligibility.
Engagement and Duties: Members are expected to actively participate in REC functions:
- They must attend scheduled REC meetings regularly. Frequent unexplained absence (e.g., missing more than four consecutive meetings without a valid reason) may lead to removal from the committee.
- Members should come prepared, having reviewed the protocols and materials distributed before the meeting.
- Each member must sign a confidentiality agreement upon appointment, committing to keep all application materials, discussion details, decisions, and other sensitive information confidential.
Termination of Membership: A member’s appointment may be discontinued if:
- Their term expires and is not renewed.
- They resign or are no longer able to serve.
- They violate confidentiality or have serious misconduct.
- They develop a conflict of interest that precludes impartial service.
- The committee (in consultation with LCPTS leadership) decides to revoke membership for reasons such as non-performance or breach of ethical standards.
Any vacancies on the committee will be filled by appointing a new member with a similar expertise profile, as needed, following the standard appointment process.
Conditions of Appointment
When considering potential REC members, certain conditions and expectations are made clear:
- Willingness to Serve: Prospective members should be willing to dedicate time and effort to REC responsibilities, including attending meetings and reviewing materials in advance.
- Formal Acceptance: An official invitation or appointment letter is provided by the appointing authority (e.g., the LCPTS Director or governing board). The appointee must indicate acceptance in writing (often by signing the letter or a memorandum of understanding).
- Public Disclosure: Members must consent to have their name, professional affiliation, and role on the REC publicly available (e.g., on LCPTS or NHRA records), as a measure of transparency.
- Compensation Transparency: Service on the REC is generally considered a public service; however, if any honorarium or reimbursement is provided (for example, for transport or meals during meetings), these transactions will be recorded. Financial records of REC-related payments are maintained and available for auditing or upon legitimate request, ensuring that no conflicts of interest arise from compensation.
Quorum Requirements
For the REC to conduct official business and make decisions on protocols, a quorum must be present:
- At least two-thirds of the committee’s voting members must be in attendance.
- The quorum must include diversity; it cannot consist exclusively of members from one profession or one gender. It also must include at least one member whose primary expertise is nonscientific (e.g., a community member or ethicist).
- If quorum is not met, the meeting cannot proceed with protocol reviews or decisions. (Administrative discussions might occur, but no official decisions on research proposals can be made.)
Committee Leadership and Roles
- Chairperson: The Chair is responsible for conducting REC meetings, guiding discussions, and ensuring that the committee’s decisions are reached in a fair and consensus-driven manner. The Chair signs off on official communications and represents the REC in dealings with researchers and institutional officials. If the Chairperson is absent for a meeting, the Vice Chairperson will assume this role. In the rare event that both are absent, the members present may elect an acting chair for that meeting.
- Vice Chairperson: The Vice Chair supports the Chair in all duties and stands in as needed. The Vice Chair may also take on special tasks as delegated by the Chair (such as overseeing subcommittees or leading certain initiatives).
- Secretary: The Secretary manages the REC’s administrative tasks. This includes scheduling meetings, preparing the agenda, distributing protocols and related documents to members ahead of meetings, and ensuring that all necessary documents are available for review. During meetings, the Secretary (or a delegated assistant) takes notes to document attendance, discussions, decisions, and dissenting opinions. After meetings, the Secretary drafts the minutes and, upon approval by the Chair, issues formal decision letters to investigators. The Secretary also maintains the REC’s archives and correspondence.
- Vice Secretary: The Vice Secretary assists the Secretary and can perform the Secretary’s duties if the Secretary is unavailable. This ensures continuity of administrative support for the REC’s functions.
All officers are expected to act impartially and uphold the REC’s integrity. They should not use their positions to unduly influence the committee’s decisions and must also recuse themselves from leadership roles in discussions where they have a conflict of interest.
Agenda Preparation, Document Distribution, and Minutes
Agenda Preparation. The Secretary drafts and circulates the agenda 7 calendar days prior to the meeting. The agenda lists: protocol IDs/titles; PI names; review type (initial/continuing/amendment/expedited report-back); assigned primary/secondary reviewers; and any conflict-of-interest disclosures received.
Distribution of Applications. Full application packets (protocol, consent/assent, instruments, IB/product info, recruitment, prior determinations) are shared with all members via the secure REC portal ≥7 calendar days before the meeting. Primary reviewers receive a “reviewer brief” highlighting key ethical considerations.
Late Materials. Revisions received <1 calendar day before a meeting are moved to the next agenda unless the Chair deems them urgent.
Minutes. The Secretary records attendance (including recusals), quorum confirmation, summary of deliberations, decisions (and vote counts if applicable), conditions required, and action items. Draft minutes are circulated to members within 5 working days; corrections are integrated and minutes are adopted at the next quorate meeting. Signed, approved minutes are archived.
Timely Notification to Members. A same-day Decision Summary Memo listing protocol outcomes is posted to the member portal (REDCap or Box) within 2 working days after the meeting for internal awareness (not a substitute for formal letters to PIs).
Training of REC Members
It is essential that REC members are well-versed in research ethics and stay current with evolving standards:
- New REC members receive initial training or orientation at the start of their tenure. This covers the REC’s SOPs, review criteria, relevant national regulations, and international guidelines.
- All members are encouraged (and may be required) to complete certified training programs in research ethics and good clinical practice. For example, members might take the online courses offered by the Collaborative Institutional Training Initiative (CITI) or similar programs, which cover topics like biomedical research ethics, social/behavioral research ethics, and regulatory compliance.
- The REC will periodically organize or recommend continuing education sessions. These could be in the form of workshops, seminars, or conferences on topics such as bioethics, regulatory updates, or specific issues like informed consent in low-literacy populations, data privacy, or animal welfare in research.
- Documentation of each member’s training (certificates, etc.) is maintained by the REC Secretariat. Members who have not kept up with required training may be reminded or, if necessary, barred from reviewing protocols until they complete the necessary training, to ensure the quality of ethics review is not compromised.
Independent Consultants
For certain research proposals, the REC may seek the assistance of independent consultants or experts to provide additional expertise:
- If a study involves highly specialized scientific content (for example, gene editing, advanced surgical techniques, or a rare disease), the REC can invite a subject matter expert to review the protocol and give an opinion.
- If a proposal raises complex ethical issues (for instance, a novel reproductive technology or a culturally sensitive intervention), the committee might consult an experienced bioethicist or a representative from the community or population in question.
- Community representatives or patient advocates may be consulted especially when a study could significantly impact a particular community or patient group.
- These consultants do not have decision-making power within the committee. They provide insight and recommendations, either through written reports or by attending the REC meeting for that agenda item. They must also agree to confidentiality regarding the information they review.
- Any honorarium or compensation for the consultant’s time is handled according to LCPTS policies and is disclosed in REC records, as transparency for any external involvement.
By leveraging independent consultants, the REC can ensure a well-rounded review of proposals that require knowledge beyond the expertise of current members.
Application Procedures and Requirements
Researchers who wish to have their study reviewed by the LCPTS-REC must prepare and submit a comprehensive application package. The submission should include all information needed for an intensive ethical and scientific review. Key requirements are:
- Cover Letter (Optional): It can be helpful to include a brief cover letter addressed to the REC Chair or Secretary, stating the intention to seek ethics approval and listing the documents enclosed.
- Completed Application Form found online: LCPTS-REC provides a standardized application form. This form captures essential details such as the project title, principal investigator’s name and qualifications, contact information, sponsor or funding source (if any), and a checklist of submitted documents. The principal investigator must sign this form (digital or ink signature as required) to affirm the accuracy of the submission.
- Research Protocol (part of the online submission): Provide the full study protocol in English. This is a detailed document describing the research question, background (with literature review), objectives, methodology, and analysis plan. The protocol should also discuss ethical considerations (risks, benefits, privacy protections, etc.). It must have a version number and date for reference. LCPTS-REC has no specific format or template, Researchers are free to use their own.
- Lay Summary (part of online submission): Include a 1 page summary of the research in non-technical language. This summary should cover the aim of the study, why it is important, what it involves for participants, and the expected outcomes. It should be understandable to someone without a scientific background.
- Informed Consent Documents (part of the online system): Drafts of all forms and scripts used in the consent process must be provided. Typically this includes:
- A Participant Information Sheet that clearly explains the study in simple terms (covering purpose, procedures, risks, benefits, voluntariness, and rights of the participant).
- A Consent Form which participants (or their legally authorized representatives) will sign to indicate informed consent. If non-English speaking populations are involved, include translations of these documents in the local language(s). (Note: The REC may require verification of translations by an independent party.)
- If applicable, assent forms for minors (children who are old enough to understand a simplified explanation of the study) along with parental consent forms.
- Study Instruments (to be included in the protocol): Append copies of questionnaires, interview guides, surveys, data collection forms, or any other instruments that will be used to interact with participants or collect data.
- Recruitment Materials: If the study will use flyers, posters, social media ads, or other recruitment tools, include samples or descriptions of these materials for the REC to review for appropriateness and accuracy.
- Investigator’s Brochure/Product Information: (For clinical trials involving drugs, vaccines, or devices) include any available information on the investigational product. This could be an investigator’s brochure, package insert, or device manual that provides safety and efficacy data from prior research.
- Evidence of Scientific Review: If the protocol has undergone scientific or technical review by another committee (e.g., a thesis committee or a scientific review board in the institution), include documentation of that review or its outcome (e.g., a letter indicating scientific approval).
- Regulatory Permissions: If applicable, attach any regulatory approvals or notifications. For example, if the study involves an investigational drug, provide evidence that the Zambia Medicines Regulatory Authority (ZAMRA) or equivalent has been notified or has approved the trial. Or if the research involves a foreign collaboration, proof of local authorizations.
- Institutional Approval: A letter of support or permission from the institution(s) or site(s) where the research will be conducted (e.g., hospital management or community leaders) should be included, if relevant.
- Budget and Funding Details: Provide a summary of the project budget and identify the funding source. This helps the REC understand the scale of the project and ensure there are no undisclosed conflicts of interest or undue influence from sponsors. If participants will be compensated, the amount and source of that compensation should be clear here.
- Researchers’ Qualifications: Include a brief CV (resume) for the Principal Investigator and other key researchers. Focus on qualifications and experience relevant to the proposed study (education, previous research, GCP training, etc.). For student projects, a CV of the faculty supervisor and a support letter from the supervisor may be required.
- Ethical Compliance Statement: A short statement within the application or protocol that confirms the investigators will comply with all applicable ethical guidelines and regulatory requirements (e.g., “We certify that this research will be conducted in accordance with the Declaration of Helsinki, CIOMS guidelines, and all applicable national regulations, and that we will promptly report any deviations or issues to the REC”).
- Conflict of Interest Declaration: Investigators should disclose any potential conflicts of interest (financial interests in a drug or device being tested, dual roles such as researcher and healthcare provider of the participants, etc.). A statement about how any conflicts will be managed should also be included.
- Other Supporting Documents: Any additional documents that add information or protection for participants. This could include:
- Data sharing agreements if the study plans to share identifiable data with external entities.
- Material Transfer Agreements if sending samples abroad.
- Community endorsement letters for community-based studies.
- Training certificates (like ethics training or GCP) for study staff.
- A schedule or timeline of the study.
All documents should be indexed or clearly labeled. The application must be submitted by the deadline specified by the REC (typically at least 5 days before a scheduled meeting). Late or incomplete submissions may be deferred to the next meeting’s agenda.
Conflicts of Interest (COI) and Conflicts of Commitment (COC) — Instructions
Scope. Applies to REC members, ad hoc reviewers/consultants, and investigators.
Member COI. A REC member has a COI if they (or immediate family): are involved in the study (PI, co-investigator, advisor), have financial interests (equity, IP, payments ≥USD 5,000/year or per LCPTS threshold), supervisory/academic authority over the PI/participant pool, or other relationships that could impair impartiality.
Member COC. A conflict of commitment exists when outside duties (consultancies, leadership roles, clinical workload) materially limit a member’s ability to fulfill REC responsibilities (e.g., chronic non-attendance, inadequate reviews, missed deadlines).
Disclosures & Management.
- Pre-meeting: Members must disclose COI/COC to the Secretary upon agenda receipt (and update at the meeting).
- In-meeting: The Chair announces declared COIs; conflicted members do not receive confidential materials beyond public info, leave the room during deliberation and voting, and are recorded as recused.
- Assignments: Conflicted members are not assigned as reviewers on conflicted items.
- COC Remedies: The Chair may reallocate workloads, issue reminders, or, if unresolved, recommend temporary suspension or replacement to maintain REC functioning.
- Records: All disclosures/recusals are minuted; a COI/COC log is maintained.
- Investigator COI: PIs must disclose study-related financial or role conflicts in the application; the REC may require a management plan (e.g., independent data review, separate clinical care vs research roles, disclosure in consent).
Consent Transparency. When relevant, the consent form must transparently disclose investigator/sponsor conflicts to participants.
Submission Procedure
To submit an application to the LCPTS-REC:
- Submission Method: Follow the instructions for submission format. Currently, applications can only be submitted electronically via email to the REC secretariat at rec@lcpts.org or via this link on our website https://lcpts.org/lcpts-rec/ . The electronic submission should be a single compiled PDF or a set of files as specified by the REC guidelines. Follow the steps on the website.
- Deadline: Although submission can be made at any given time, researchers are encouraged to take advantage of the submission deadlines relating to REC review meetings. Generally, the REC meets monthly, and protocols should be submitted at least 2-5 days prior to the meeting at which review is desired. Submissions after the cut-off date will be held for the subsequent meeting. However, meetings will still be called upon by the chair or vice chair to review and attend to outstanding submissions weekly.
- Confirmation: Once submitted, the REC office will acknowledge receipt of the application (usually via email, indicating if the submission is complete or if any documents are missing).
- Distribution to Members: The REC Secretary will distribute the application materials to REC members (or assigned reviewers) ahead of the meeting, giving them adequate time to review.
- Coordination: If the Principal Investigator is not based in Zambia or at LCPTS, it is required to have a local co-investigator or collaborator who can liaise with the REC. This local researcher’s name and contact information should be provided, ensuring that the study has local oversight and accountability.
- Fee Payment: Ensure that any required review fees are paid. The current fee structure will be provided by the REC office. Proof of payment (such as a deposit slip or receipt) should accompany the application. The REC will not issue an approval until all fees are settled.
Researchers are encouraged to contact the REC office or Secretary for any clarifications on submission requirements before submitting, to avoid delays due to administrative issues.
Review Procedure
Once a submission is received and deemed complete, it undergoes the following review process:
- Pre-review (Administrative Review): The REC Secretary (or an assigned administrator) will first check the submission for completeness. If documents are missing or if any obvious issues are noted (e.g., unsigned application form, missing pages), the investigator will be asked to correct these before the ethical review proceeds.
- Assignment of Reviewers: The REC Chair or Secretary will assign primary reviewers for the protocol. Typically, two members with relevant expertise are appointed to thoroughly review the submission and present their findings. For instance, if the study is a clinical trial, a clinician and a biostatistician might be primary reviewers; if it’s a social science study, a social scientist and an ethicist might be assigned.
- Distribution of Materials: All REC members receive at least a summary of the protocol and consent materials, while primary reviewers receive the full packet. Members are expected to review in advance, but primary reviewers will lead the discussion.
- Meeting Discussion: During the REC meeting, the Chair will introduce each protocol on the agenda. The primary reviewers will present their assessment, noting any ethical issues, queries, or recommendations. This is followed by an open discussion where other members can ask questions or express their opinions.
- Investigator Presence: Sometimes, the REC may invite the investigators (or their representatives) to present the study or answer specific questions at the beginning of the review discussion. If invited, the investigator will provide a brief summary and clarifications. However, investigators are not present for the committee’s private deliberations and decision-making to maintain independence of the review.
- Decision Making: After discussing, the REC arrives at a decision (as detailed in the next section on Decision Making). If consensus is challenging, a vote may be taken.
- Minutes: The Secretary documents key points of the discussion and the decision reached for each protocol. Any abstentions or recusals (and their reasons) are noted in the minutes.
- Communication of Outcome: Following the meeting, the REC Chair/Secretary will prepare an official letter to the investigator summarizing the decision and any required actions or recommendations (see Communicating Decisions section for details).
- Timelines: The typical time from submission to receiving a decision can vary. Generally, if a protocol is submitted by the deadline and is straightforward, a decision letter is issued within 1-2 days after the meeting. Complex cases or those requiring clarifications may take longer due to the need for additional information and possibly an extra review cycle. The REC strives to make the process efficient while ensuring thorough review.
If a research proposal requires especially urgent attention (for example, a study related to an ongoing disease outbreak), the applicant can request an accelerated review or the REC can convene an emergency meeting (see Expedited Review).
Suggested timelines
- Assignment: Chair/Secretary assigns primary and secondary reviewers within 1-2 working days of docketing.
- Member Access: Full packets available ≥2-5 days before the meeting via secure portal.
- Reviewer Reports: Primary reviewers submit written comments within ≥1-5 working days before the meeting to the Secretary for compilation and circulation to all members.
Meeting Cadence
Frequency of Meetings. The LCPTS-REC convenes once per month for a full-board meeting (standard: the first Thursday of each month, 14:00–17:00 CAT). Additional ad hoc or extraordinary meetings may be called by the Chair for time-sensitive reviews (e.g., outbreaks, national priority studies) or to manage heavy loads. These meetings may be held weekly.
Annual Calendar. The Secretary circulates an annual meeting calendar by 15 January each year and posts it on the REC intranet, whatsapp group, email group or in Box.
Submission Cut-offs. Complete applications must be received ≥2-5 calendar days before a scheduled meeting to be docketed for that agenda.
Elements of Review
In evaluating each research proposal, the REC focuses on several key aspects to ensure both ethical acceptability and scientific merit. The following elements are scrutinized:
Study Design and Conduct
The committee assesses the scientific soundness and statistical validity of the study, as well as its ethical design:
- Scientific Merit: Does the study have a clear research question based on sound background and justification? A thorough literature review should be provided to demonstrate the need for the study. The design (e.g., randomized trial, observational study, qualitative interviews) should be appropriate to answer the question.
- Methodology: Are the procedures and methods described clearly and in detail? This includes sampling methods, interventions, data collection procedures, and analytic plan. Adequate information about sample size calculation should be given to ensure that the study is neither too small (to be futile) nor excessively large (exposing more participants than necessary).
- Risk-Benefit Analysis: What are the potential risks (physical, psychological, social, or economic) to participants, and what benefits are expected (to participants or society)? The REC considers whether risks have been minimized and are reasonable in relation to the anticipated benefits. For example, invasive procedures or tests should only be done if crucial to the objectives, and safety monitoring plans should be in place for higher-risk studies.
- Participant Selection (Justice): Are the inclusion and exclusion criteria fair? The REC ensures that the selection of subjects is equitable and justified by the scientific goals—not exploiting or excluding groups without cause. If the research targets a specific group, is this targeting ethically justified? Also, the REC checks that vulnerable groups are not included unless necessary (and then, with special protections).
- Stopping Rules and Withdrawal: The protocol should explain under what conditions participants will be withdrawn or the study terminated early (e.g., severe adverse events, interim results showing efficacy or futility). This ensures there is a plan to protect participants if things go wrong or if new information emerges.
- Adequacy of Facilities: The REC looks at whether the research site has the capacity to conduct the study safely. For clinical studies, is there access to emergency care and necessary medical expertise? For lab research, are biosafety measures in place if dealing with hazardous materials?
- Research Team: The qualifications and experience of the principal investigator and study team are reviewed. Do they have the expertise to carry out the study and handle potential complications? If the study spans multiple disciplines, are appropriate collaborators on board? The REC may require evidence of GCP training for clinical trials or other specialized certifications as relevant.
- Data and Safety Monitoring: For interventional studies, especially clinical trials, is there a Data Safety Monitoring Board (DSMB) or an equivalent safety monitoring plan outlined? The REC considers whether an independent oversight of data and safety is warranted and whether it has been addressed.
- Result Dissemination: The protocol should indicate plans for disseminating results (e.g., publication, report to stakeholders). While not an approval requirement per se, transparency and commitment to share results (including negative or inconclusive findings) are ethically encouraged.
Recruitment of Participants
The strategies for identifying and recruiting participants are reviewed to ensure they are ethical and respectful:
- Recruitment Methods: How will potential participants be approached? The REC evaluates recruitment materials (flyers, social media posts, verbal scripts) to ensure they are not misleading or coercive. Recruitment should be done in a manner that respects privacy; for instance, obtaining permission before contacting individuals referred by third parties.
- Study Population: The demographics of the intended participants (age, gender, socio-economic status, etc.) are considered. The REC checks if the population is appropriate for the research question and if any group is unfairly targeted or excluded. For example, if a study focuses on a disease prevalent in both men and women, excluding one gender needs a valid reason.
- Vulnerability and Consent Capacity: Are participants likely to include people who might have limited capacity to consent (e.g., children, mentally impaired individuals)? If so, additional safeguards must be in place.
- Inclusion/Exclusion Criteria: The criteria for who can or cannot participate are reviewed to ensure they are justified. Sometimes criteria that seem restrictive are necessary for safety or scientific clarity, but the REC verifies this. Unnecessary exclusion of groups (like women of childbearing age, older adults, etc.) is questioned unless justified.
- Training of Recruiters: Those obtaining consent or recruiting participants should be adequately trained. If the study involves sensitive topics or vulnerable groups, the recruiters/interviewers should have specialized training (for instance, counseling skills for sensitive interviews).
- Compensation Disclosure: If there is any compensation or incentive for participation, the recruitment process must clearly inform potential participants of this without emphasizing it in a coercive way. The REC checks that the amount is appropriate (not too high to constitute undue inducement).
Care and Protection of Participants
The protocol must demonstrate respect and care for the participants’ well-being throughout the study:
- Informed Communication: How will information be conveyed to participants during the study? Beyond the initial consent process, the REC looks for plans to keep participants informed, for instance, providing them with any new findings that might affect their willingness to continue, or simply keeping them updated on study progress if appropriate.
- Medical Care: For studies that involve any risk of medical complications or injury, the REC ensures that there is a plan for medical care. For example, if a participant has an adverse reaction, is there immediate care available and who covers the costs? If the study involves a therapeutic intervention, the REC asks what care is provided to participants (during and, if needed, after the study).
- Standard of Care Considerations: If the study’s methodology requires withholding standard therapy (e.g., a placebo-controlled trial where an existing effective treatment exists), the REC weighs this carefully. Such a design must be justified by a genuine scientific necessity and minimized in duration/extent. Participants must be fully informed about what is being withheld and any additional monitoring or rescue mechanisms must be in place. In many cases, the REC might require that any participant who deteriorates be taken off the placebo or provided treatment.
- Withdrawal Implications: The committee ensures participants are free to withdraw at any time. The protocol should clarify that choosing to withdraw will not compromise the participant’s subsequent medical care or any other entitlement. It should also specify what will happen to data or samples already collected if a participant withdraws (usually, data up to the point of withdrawal is retained for integrity unless the participant requests otherwise).
- Compensation for Harm: If there is a risk of injury or harm, the REC checks that the consent form and protocol clearly state what compensation or care will be provided. For instance, if a trial participant is injured by the study intervention, will the sponsor or institution cover the medical costs? The absence of a fair policy for compensation is a serious ethical concern.
- Post-Trial Access: In studies where an intervention is found to be beneficial (e.g., a drug that works or a successful intervention program), the REC considers whether and how participants (or the community) can continue to access it after the research is over. If the sponsor or investigator has made commitments regarding this, those should be documented. For example, providing a successful drug to participants until it’s available on the market.
- Participant Costs: The protocol and consent form should outline any costs participants might have to bear (travel, time off work, etc.) and whether the study will cover/reimburse those. The REC favors studies that try to minimize out-of-pocket costs for participants, to avoid economic barriers to participation.
- Participant Compensation: If there are payments or gifts to participants, the REC evaluates them to ensure they are reasonable. Compensation should typically cover expenses and time, and perhaps an inconvenience allowance, but not be so high as to unduly influence someone to take risks. Any such compensation must be clearly communicated in the consent process.
Confidentiality
Protecting the privacy of participants and the confidentiality of research data is paramount:
- Data Protection Measures: The REC reviews how the study will collect, store, and secure data. Will identifiable information be coded or anonymized? Who will have access to the key linking codes to identities? Data should ideally be stored in locked cabinets or secure electronic databases with password protection.
- Use of Personal Data: If personal medical records or records from other institutions will be accessed, the REC ensures the protocol has permission to do so and that participants consent to this access. Only data necessary for the research should be collected (data minimization principle).
- Privacy in Procedures: Consideration is given to whether any study activities could intrude on privacy (e.g., home visits, interviews on sensitive topics) and if so, how the study team will handle these sensitively.
- Genetic or Stored Samples: If DNA analysis or future unspecified research on samples is planned, confidentiality issues become even more critical. The REC looks at whether genetic information might have implications for privacy of the individual or their family (for example, in revealing predispositions to disease) and whether appropriate counseling or confidentiality protections are in place.
- Legal Compliance: The REC will ensure that the research complies with any data protection laws (for example, any local data privacy statutes). Cross-border data transfer (sending data to another country) must also meet legal requirements and typically requires explicit participant consent and assurances of equivalent data protection in the receiving country.
Use of Biological Specimens
When research involves taking biological samples from participants:
- Scope of Sampling: The types and amounts of specimens (blood volume, number of biopsies, etc.) must be clearly described and justified. The REC checks that the frequency and quantity of sampling are not excessive or harmful.
- Purpose of Use: The protocol should state what the samples will be tested for in the current study. If any tests might reveal incidental findings (like a health condition unrelated to the study aims), the plan for handling that (including whether the participant will be informed) should be addressed.
- Storage and Future Use: If samples will be stored beyond the study for future research, this needs to be stated in the consent form, including for how long and under what conditions (identifiable, coded, or completely anonymized). The REC ensures that consent for future use is specific enough (or that separate consent will be obtained for future studies) and that there is a plan for storage security. Long-term storage might require additional regulatory approvals in some cases.
- Export of Samples: When researchers plan to send samples abroad for analysis, the REC will verify that this is disclosed in the consent form and that appropriate approvals (such as export permits or MTAs) are or will be in place. Samples should not be sent to another country without confirming compliance with both local and international requirements.
- Benefit Sharing: In some cases, if commercial products or patents could result from the use of human biological materials, the REC may consider whether the consent process addresses this (though specifics are often hard to predict, transparency with participants is encouraged if it’s a possibility).
- Sample Disposal: The protocol should explain what happens to samples at the end of the study or if a participant withdraws consent for use of their samples. Typically, participants can choose to have remaining identifiable samples destroyed if they withdraw. Otherwise, at study end, samples might be destroyed or moved to a biobank with consent. Proper disposal methods (incineration for biohazard, etc.) should be used and documented.
Informed Consent Process
The REC examines the consent process in detail to ensure it is ethically and legally sound:
- Process and Setting: Consent should be obtained in a setting that allows privacy and enough time for discussion. The REC checks if the protocol describes when and where consent will be sought (e.g., in a quiet room at a clinic, not at a crowded registration desk) and that participants won’t be rushed or coerced.
- Who Obtains Consent: The qualifications of the person obtaining consent are considered. Ideally, it should be someone knowledgeable about the study and able to answer questions, often a member of the research team, but not someone in a position of authority over the participant (to avoid undue influence; e.g., a doctor who is also the participant’s personal physician might not be ideal for obtaining consent to a risky trial they are running).
- Voluntariness: The consent materials must emphasize that participation is voluntary. The REC ensures there are no statements that waive the participant’s legal rights or that appear to release the researchers or sponsors from liability for negligence (such waivers are not permitted).
- Language and Length: The language level should be simple enough that an average person with basic education can understand. Technical terms should be explained or avoided. The REC often has a guideline (such as ~8th grade reading level) and might use tools or check readability. They also prefer shorter, to-the-point consent forms (covering all key information ideally within a few pages). If a consent form is very lengthy due to complex study information, the REC may suggest including a simpler summary or at least ensuring the main points are prominently highlighted.
- Key Information Covered: The REC verifies that the consent form covers all required information:
- Purpose of the research, and a statement that it involves research (so participants know it’s not part of standard care, if it’s a clinical study).
- What procedures will be done and which aspects are experimental or beyond routine care.
- Foreseeable risks or discomforts, and efforts taken to minimize them.
- Expected benefits (if any) to the participant; if no direct benefit, it should be stated clearly (while possibly noting potential benefits to others or to scientific knowledge).
- Disclosure of appropriate alternative procedures or courses of treatment, if the participant chooses not to participate (applicable in therapeutic research).
- A statement about the extent to which confidentiality of records will be maintained (who might inspect the records, etc.).
- For research involving more than minimal risk, an explanation of whether any compensation or medical treatments are available if injury occurs, and if so, what they consist of or where further information can be obtained.
- Contact information for the researchers (for questions about the study) and for an independent contact (often the REC or a patient ombudsman) for questions about rights or to report concerns.
- A statement that participation is voluntary and that refusal to participate or withdrawal will involve no penalty or loss of benefits to which the participant is otherwise entitled.
- Consent for Special Situations: If any participants cannot physically sign or are illiterate, the method for documenting consent (a thumbprint, an impartial witness signature confirming the person understood and consented, etc.) should be described. The REC may require a witness line on the consent form in such cases.
- Re-consent: The committee looks at whether any situations would require re-consenting participants. For example, if the study protocol changes significantly, or if new information emerges about risks, participants should be informed and their consent obtained again. This plan should be in the protocol.
- Document Retention: Investigators must keep the signed consent forms on file (usually for at least the duration of the research and as mandated by regulations, often several years after). The REC ensures that participants also get a copy of the consent form for their own records.
- Translations: Where the study population includes non-English speakers, all consent documents should be translated into the appropriate local language(s). The REC may require proof that the translation is accurate (for instance, a back-translation or a certificate by a translator).
- Continuous Consent: Especially in longer studies, the consent process is not a one-time event but ongoing. The REC encourages investigators to continue the dialogue with participants, checking understanding and willingness to continue at various points, and promptly answering any new questions that arise as the study progresses.
- Complaint Mechanism: Participants should be told how they can contact someone independent of the research team (e.g., an REC representative or an ombudsperson) if they have concerns or complaints about the study. This provides a mechanism for participants to report issues. The REC makes sure the contact provided is functional and will be responsive to participant inquiries or grievances.
Community Considerations
For studies conducted in community settings or that significantly impact communities:
- Community Engagement: The REC looks for evidence that the researchers have engaged or plan to engage with the community from which participants will be drawn. This could mean consulting community leaders, holding community meetings, or involving community advisory boards. Such engagement can build trust and ensure the research is culturally appropriate.
- Community Benefit: The potential benefits of the research to the community should be considered. Will the community gain knowledge, access to interventions, or other direct benefits from the study? If the community is bearing any burden of research, fairness dictates they should stand to benefit from or at least not be harmed by the results.
- Cultural Sensitivity: The design and consent process should respect local cultural norms and values. For example, in some communities, it’s appropriate to seek permission from community elders or to explain the study in a community gathering. The REC will consider whether these cultural steps, where appropriate, have been taken (so long as they do not override individual consent).
- Impact on Community: Could the research adversely affect the community’s standing or cause stigma? For example, publishing that a certain community has a high prevalence of a disease could potentially stigmatize it. The REC may require that the community be informed of results in a sensitive manner and that steps are taken to mitigate any possible negative fallout.
- Post-research Feedback: A plan to communicate results back to the community is encouraged. This might be through community meetings, reports or leaflets in the local language, or other culturally appropriate ways. The REC sees this as part of showing respect to the community and fulfilling the ethical obligation of disseminating findings.
- Capacity Building: If the research involves the local healthcare infrastructure or personnel, does it help build capacity? For instance, are local health workers being trained as part of the project? Are there improvements to local facilities (even temporary) that could have lasting benefits? The REC views favorably those projects that strengthen local capacity or knowledge.
- Affordability and Accessibility: If an intervention being tested is likely to be beneficial, the REC may ask whether there’s consideration for making it accessible to the population after the study. It’s understood this is often beyond the researchers’ control (policy or market decisions), but early consideration (like involving health authorities if a drug trial is successful) is encouraged.
Vulnerable Populations
Special care is taken if the research involves populations that may have limited ability to give informed consent or are at higher risk of coercion or undue influence:
- Identifying Vulnerability: The protocol should identify if participants include, for example, children, pregnant women, prisoners, people with cognitive impairments, economically disadvantaged individuals, refugees, or the elderly. Each of these groups has specific ethical considerations (e.g., regulations often limit research on pregnant women unless for their or the fetus’s benefit; prisoners require extra oversight to ensure participation is truly voluntary and not linked to any prison benefits).
- Additional Safeguards: The REC ensures that the study describes safeguards like obtaining consent from legal guardians for those who can’t consent, ensuring that participation (or non-participation) will not affect entitlements (for instance, a prisoner’s participation will not influence their parole or privileges), or that the research is directly relevant to the vulnerable population’s needs.
- Risk Limits: For some vulnerable groups like children, regulations often categorize research by risk level (minimal risk, minor increase over minimal risk with direct benefit, etc.). The REC will apply these criteria to decide if the research is permissible. Research with no prospect of direct benefit to a child, for example, must generally not exceed minimal risk or a minor increase over it.
- Assent: For those unable to give full consent (like older children), the REC looks for an assent process where the individual’s agreement is also sought to the extent of their capability, in addition to consent from a parent or guardian.
- Protection from Exploitation: The REC is alert to situations where vulnerable people might feel pressured to join (for example, students might feel they must participate in a professor’s study or patients might feel their care will be compromised if they don’t join a doctor’s research). The protocol and consent process should make clear that services or grades or care will not be affected by the decision to participate or not.
- Privacy for Vulnerable Groups: Some populations (like those with certain medical conditions or those who are illiterate) need particular care in the consent process to avoid embarrassment or breach of privacy. The REC appreciates when researchers tailor their approach (e.g., using pictures or verbal consent processes for those who cannot read, having an independent person ensure a prisoner’s consent is voluntary, etc.).
- Regulatory Compliance: The REC will ensure that any research involving vulnerable groups complies with all relevant regulations. For instance, international standards (such as U.S. 45 CFR 46 Subparts B, C, D for pregnant women, prisoners, children) provide specific protections, and the REC will be guided by such standards in addition to local regulations.
Animal Welfare Considerations
For research that involves animal subjects, the REC (which in this context functions similarly to an Institutional Animal Care and Use Committee) will evaluate:
- Rationale for Animal Use: The protocol must clearly justify why animals are needed and why alternative methods (in vitro experiments, computational models, or use of less sentient organisms) cannot answer the research question (Replacement principle).
- Species and Number of Animals: The choice of animal species should be appropriate for the scientific objectives, and the number of animals should be the minimum required to achieve statistically valid results (Reduction principle). An explanation of how the numbers were determined (e.g., via power analysis or experience from previous studies) should be provided.
- Housing and Care: Details about the housing, feeding, and care of the animals should be included. Facilities must meet standards for humane housing (proper ventilation, bedding, opportunities for species-specific behavior, social housing for social species, etc.). Veterinary care should be available, and routine observation should be in place to monitor animal health.
- Procedures and Pain Management: Any procedures that might cause pain or distress must include descriptions of how pain/distress will be minimized (Refinement principle). This includes the use of anesthesia or analgesics for surgical or potentially painful procedures, humane endpoints (criteria for when an animal will be removed from the study or euthanized to prevent suffering), and ensuring personnel are trained in proper handling to reduce stress.
- Ethical Compliance: The research should comply with relevant animal research ethics guidelines and laws. For instance, researchers should follow the Guide for the Care and Use of Laboratory Animals and adhere to the principles of the 3Rs (Replacement, Reduction, Refinement) for humane animal experimentation. If Zambia has specific legislation or standards for animal research, those must be observed, and necessary permits obtained.
- IACUC Approval: If LCPTS or a collaborating institution has a dedicated Animal Ethics Committee or IACUC, evidence of that committee’s approval should be provided. If not, the LCPTS-REC will itself ensure a thorough ethical review of animal use. The REC may include or consult a veterinarian or animal care expert during the review of such protocols.
- Disposal or Post-Research Use: The protocol should state what happens to the animals after the study. If euthanasia is planned, it should be done humanely in accordance with accepted guidelines (such as those of the American Veterinary Medical Association or international standards). If animals will remain alive, plans for their care or adoption should be described.
- Training of Personnel: All personnel handling animals must be appropriately trained in animal care and use. The REC may ask for proof of training or qualifications for specific procedures (e.g., surgical techniques on animals, animal anesthesia).
- Monitoring: Just as with human studies, animal studies should have monitoring in place. This could be daily health checks with logs, and predetermined criteria for intervention if an animal’s condition deteriorates. Any unexpected animal welfare issues or higher-than-anticipated morbidity/mortality must be reported to the REC promptly, and the REC can require modifications or halt the study if serious welfare concerns arise.
By reviewing these factors, the REC ensures that animal research is justified and conducted to high standards of humane care, balancing scientific objectives with ethical responsibility toward animal subjects.
Expedited Review
Under certain conditions, the LCPTS-REC offers an expedited review process, sometimes referred to as a “fast-track” review:
- Eligibility for Expedited Review: Studies that present no more than minimal risk to participants and involve procedures falling within categories recognized as eligible for expedited review (for example, anonymous surveys, non-invasive sample collection, analysis of existing data, minor amendments in approved studies, etc.) can be considered for expedited review. Additionally, when timing is critical (such as during public health emergencies or for nationally important research that needs quick initiation), the REC may use an expedited process.
- Process: When a researcher requests expedited review (or the REC deems it appropriate), the REC Chair will appoint a subset of REC members (usually 2 to 3 experienced reviewers) to evaluate the protocol outside of a full committee meeting. These reviewers will independently review the submission and then confer to reach a decision.
- Timeline: Expedited reviews are conducted more rapidly than full reviews. The REC aims to provide a decision or feedback within a shorter window (for instance, within 1 week of submission, or even faster if urgent). In some cases, initial feedback might be given within 72hrs.
- Possible Outcomes: The designated expedited reviewers can recommend the same decisions as the full committee (approval, conditions requiring modification, deferral to full board, or rejection). If they have minor queries or require clarifications, they may communicate with the investigator via the REC Secretary to resolve them quickly.
- Limits of Expedited Authority: If the expedited reviewers feel that the protocol is more complex or risky than initially thought, they can refer it to the next full REC meeting instead of making a final decision. Also, they typically do not outright reject a study via expedited review; if they believe it should be disapproved, that decision is usually confirmed by the full committee.
- Communication and Transparency: Any proposal approved (or otherwise decided upon) by expedited review will be reported to the full REC at its next meeting. This ensures that all members are aware of expedited decisions and can provide input retroactively if needed. The decision letter to the researcher will clearly indicate that the study was reviewed through an expedited process.
- Documentation: The same documentation requirements apply to expedited reviews as to full reviews. The reviewers’ comments and the decision are recorded and filed. If conditions were imposed via expedited review, the follow-up of those conditions (and their resolution) is tracked similarly to a full board review.
Expedited review is a mechanism to increase efficiency but not at the expense of rigor. The ethical standards of review remain the same as for a full committee review; only the process timeline is accelerated.
Decision Making
After review, the committee will come to one of the following decisions for each protocol:
- Approved: The research protocol is approved as submitted (or with only very minor administrative corrections). The investigator will receive an approval letter and can proceed with the study once any administrative requirements are fulfilled (such as institutional permissions or fee payments).
- Conditional Approval (Modifications Required): The REC finds the study in principle to be approvable, but certain issues must be addressed before final approval is granted. These could be clarifications, consent form wording changes, additional risk mitigation measures, or other protocol adjustments. The committee’s letter will detail the required modifications. The researcher must resubmit the revised documents addressing these points. If the corrections are verified (often by the Chair or a subcommittee) as satisfactory, an approval will be issued. The study cannot start until the conditions are met and final approval is given.
- Deferred (Tabled): The REC is unable to make a decision due to significant questions or the need for major clarifications/additional information. The protocol is not rejected outright, but the review is postponed. The committee will provide feedback outlining what information or changes are needed. The researcher must respond to these points and the revised or supplemented application will be reviewed at a subsequent meeting.
- Rejected (Not Approved): The REC does not approve the research. This happens when the committee identifies fundamental ethical or scientific flaws that make the study unacceptable (e.g., an unfavorable risk-benefit ratio, serious ethical violations, or infeasible study design). The decision letter will explain the reasons for rejection. The researcher may choose to substantially redesign the project and submit it as a new proposal in the future, or they may appeal the decision if they believe it was based on incorrect or incomplete information (see Appeal Process below).
- Referred to National Ethics Board: In some cases, the REC may decide that a proposal should be reviewed at the national level (NHREB) rather than locally — especially if it involves sensitive issues of national concern or falls outside the REC’s authorized mandate. In such cases, the REC will forward the application to the NHREB (possibly with the local REC’s comments) and inform the researcher of this referral.
Decision Process and Ethics:
- All decisions are made during quorate meetings (see quorum requirements above). Every effort is made to reach a consensus; if a vote is needed, a majority of those present will decide, and the voting outcome is recorded in the minutes.
- REC members who have a conflict of interest with a proposal (for instance, if a member is an investigator on the study, or a close collaborator of the investigator, or has a financial interest in the outcome) must declare this conflict and recuse themselves. They will leave the room during discussion and voting on that item. This recusal is recorded in the meeting minutes to maintain transparency.
- Independent consultants or guests (if present to provide expertise on a protocol) do not vote on the decision. They exit or remain silent when the committee deliberates on final decision-making.
- If the REC issues a conditional approval or deferral, the correspondence will outline exactly what is required for approval, so the researcher knows how to respond. The committee will also decide the process for verifying the response (for example, review by the Chair or a small subcommittee vs. bringing the revised protocol back to the full committee).
- For conditional approvals where only non-controversial changes are needed (such as fixing consent language or adding a missing document), the REC often empowers the Chair (and/or a delegate such as the Vice Chair or Secretary) to issue final approval once the corrections are made, rather than waiting for another full meeting. This streamlines the process while still ensuring the conditions are met.
- In cases where major changes are made to a protocol in response to REC feedback, the REC may request a full re-submission for review to ensure all issues are adequately resolved.
- Appeal Process: If a researcher disagrees with an REC decision (e.g., a rejection or certain required changes), they may appeal. The appeal should be in writing, addressing the committee’s concerns or providing additional justification or information. The REC will re-evaluate the decision in light of the appeal. This may involve seeking external expert opinion or referring the matter to a higher institutional authority or the NHREB for independent review. The outcome of an appeal (whether the decision is upheld or modified) will be communicated in writing and is generally considered final for that submission.
All decisions and their justifications are documented in the meeting minutes and communicated to the investigator in writing.
Timeliness Standards — Internal Service Levels (SLAs)
- Completeness Check: within 1 working day of receipt.
- Routing to Reviewers: within 1 working day of docketing.
- Post-meeting Decision Letters to PIs: within 1-2 working days.
- Expedited Reviews: initial feedback within 72 hours; final letter within 5 working days of receiving satisfactory clarifications.
- Member Decision Summary Memo: within 2 working days post-meeting.
Communicating Decisions
Timely and clear communication of the REC’s decision is crucial:
- Timeline: The REC aims to send out decision letters within 1-2 days of the meeting at which the study was reviewed (or of the expedited review decision). This ensures investigators are informed promptly.
- Format: The decision is usually communicated via an official letter on LCPTS-REC letterhead (often via email, with a hard copy available on request). The letter is addressed to the Principal Investigator and copied to other relevant parties as appropriate (such as co-investigators, a student’s academic supervisor, institutional research office, or sponsor).
- Contents of Decision Letter: The letter will include:
- The title of the study and a reference number or identifier (if the REC assigns an ID to each submission).
- The date of the REC meeting when the review took place (or the date of the expedited review decision).
- The name of the principal investigator (and the affiliated institution, if different from LCPTS).
- A listing of the documents that were reviewed (e.g., protocol version X dated Y, consent form version X, questionnaires, etc.), to ensure clarity on what was evaluated.
- The decision outcome, stated clearly (e.g., “Approved”, “Approved with Conditions”, “Deferred – clarify and resubmit”, “Not Approved”).
- For approved studies: any specific remarks or reminders. This typically includes the approval’s effective date and duration (many RECs grant approval for one year at a time for long studies), and investigator obligations such as: submitting progress reports at designated intervals, reporting serious adverse events, seeking approval for any protocol amendments, and submitting a final report at the study’s conclusion. The letter may require the investigator to formally acknowledge and accept these conditions (sometimes by signing and returning a copy).
- For conditionally approved or deferred studies: a detailed list of the required modifications or additional information. Each required change or question will be enumerated. Instructions will be given on how to submit responses (e.g., provide a revised protocol with changes highlighted, a memo responding to each point) and whether the response will undergo expedited review or be tabled for the next meeting. A timeframe for responding might also be indicated (commonly, investigators are asked to respond within a certain number of weeks or months).
- For rejected studies: the reasons for the decision will be clearly explained. This can be formatted as a list of the major concerns or deficiencies that led to disapproval, referencing ethical principles or guidelines where relevant. The investigator is usually informed of the opportunity to address these concerns and reapply, or to appeal if they believe an error in the review has occurred.
- Contact information for further inquiries (usually the REC Secretary or administrative office).
- The signature (or name/title) of the REC Chairperson (or an authorized representative such as the Vice Chair or Secretary), and the date of issuance of the letter.
- Documentation: A copy of the decision letter is kept in the REC’s file for that protocol and logged in the outgoing correspondence. Any confirmation of receipt from the investigator is also kept.
- Regulatory Notification: The REC will notify relevant regulatory bodies of its decisions as required by law or policy. In Zambia, for instance, the NHRA requires that all approved research be registered with them; often the investigator is responsible for this registration using the REC approval letter, but the REC may also share periodic reports of approved protocols with the NHRA.
- Communication with Institutional Leadership: LCPTS management (such as the Center Director or research coordinator) is kept informed about the REC’s decisions. For example, summaries of protocols approved or rejected may be included in internal reports. This helps the institution support approved projects and be aware of any issues.
- To Investigators (PIs): Formal decision letters are issued within 1-2 working days of the meeting/expedited decision.
- To REC Members: Decision Summary Memo (above) within 2 working days; full signed minutes at next meeting (draft within 5 working days).
- To Regulators/Institution: Where required, notifications or registers are updated within 10 working days.
The communication process ensures that investigators are fully informed of the outcome and any next steps, and it creates a formal record of the REC’s decisions for accountability.
Follow-up Procedures
Definitions.
- SAE: Any event that results in death, is life-threatening, requires or prolongs hospitalization, results in significant disability/incapacity, or a congenital anomaly/birth defect.
- UPIRTSO: Unanticipated Problem Involving Risks to Subjects or Others (increased risk, unexpected in nature/frequency/severity, related or possibly related to participation).
Investigator Reporting Timelines to REC.
- SAE that is fatal or life-threatening: initial notice within 24 hours, detailed written report within 7 calendar days.
- All other SAEs and UPIRTSOs: written report within 7 calendar days of awareness.
- Safety updates/DSMB reports: submit within 10 working days of receipt.
REC Actions. The Chair (or designee) conducts an initial risk assessment within 2 working days; the REC may require protocol modifications, pauses, re-consenting, or increased monitoring, and will notify the PI in writing. The REC informs NHRA/regulators per national requirements.
Annual/Continuing Review Safety Summary. PIs must include cumulative AE/UPIRTSO summaries and risk re-assessment with each continuing review.
Ethical oversight continues after a project is approved. The LCPTS-REC requires and undertakes several follow-up activities:
- Progress Reports: Investigators must submit periodic progress reports on their approved research. The frequency (e.g., every 6 months or annually) will be specified in the approval letter or REC policy. These reports should include the status of study activities (number of participants enrolled vs. expected, any withdrawals), a summary of any adverse events or unexpected problems, any interim results if available, and confirmation that the study is following the approved protocol (or noting any minor deviations). The REC reviews these reports to ensure continued ethical compliance and may request additional information or action if concerns arise.
- Renewal of Approval (Continuing Review): For studies that extend beyond the initial approval period (often two years), the REC will conduct a continuing review. The investigator will be asked to submit a Continuing Review application (which may be similar to a progress report but with more detail on risk assessment and any new literature). The REC can then extend approval for another period. If a continuing review is not submitted on time, the study’s approval may lapse, and all research activities should pause until re-approval is granted.
- Site Monitoring: The REC or its authorized representatives may conduct monitoring visits or audits of the research project. This can be random or triggered by a concern. Monitoring could include reviewing informed consent records to ensure forms were properly signed, checking data privacy measures, or observing the consent process. Investigators are expected to cooperate with any such audits. These visits help verify that the study is being conducted as approved and that participants are indeed protected.
- Reporting of Serious Adverse Events (SAEs) and Unanticipated Problems: The investigator is required to promptly report any SAE or unanticipated problem involving risks to participants or others. The SOP should define the timeline (e.g., within 24-72 hours for fatal or life-threatening events, and a written detailed report within a week). The REC will review these reports and may require actions such as halting the study, modifying the protocol, informing current participants, or other safety measures. The REC may also need to report these to regulatory authorities.
- Protocol Deviations/Violations: If the research team deviates from the approved protocol without prior approval (except to eliminate immediate hazards to a participant), this must be reported. The report should explain the deviation, why it happened, and what corrective action will prevent future occurrences. The REC takes note of these and determines if the deviation compromised participant safety or data integrity and if further action is needed. Serious or repeated violations might lead to suspension of the study’s approval.
- Amendments: Any changes the investigator wishes to make to the protocol or consent materials must be reviewed and approved by the REC before implementation (except when necessary to protect participants in an emergency). Examples include changes in procedures, addition of new study sites, changes in sample size, or alterations to eligibility criteria. The investigator submits an amendment request describing and justifying the changes, along with revised documents. The REC reviews the amendment (possibly expedited if changes are minor). Only upon receiving an approval letter for the amendment can the researcher implement the changes. All amendments (approved or not) are documented in the study’s file.
- Early Termination or Completion: If a study ends earlier than expected (whether due to interim results, futility, safety concerns, or other reasons), the investigator must inform the REC and provide reasons. Likewise, at the planned end of the study, the investigator should notify the REC that the study is complete. In both cases, a final report is generally required. The final report includes number of participants enrolled, a summary of results (if available), and any issues encountered. This allows the REC to close its active monitoring of the project.
- Post-Study Obligations: Even after data collection is done, if the analysis or findings reveal something that impacts participant welfare (for example, a genetic finding important to a participant’s health), the REC may advise the researcher on ethical obligations to communicate such information. Similarly, if results are published, the REC appreciates receiving copies or references.
- Participant Complaints or Feedback: If a research participant or someone in the community raises a concern or complaint about the study (e.g., feeling pressured to join, not being treated respectfully, breach of privacy, etc.), the REC should be informed (participants are given contact info as part of consent). The REC will investigate the complaint, which may involve talking to the participant, the research team, and reviewing records. Depending on the findings, the REC can recommend or mandate corrective actions. This could include re-training the research staff, revising procedures, or in severe cases, suspending approval. The outcome of such inquiries is communicated to the complainant and recorded.
- Suspension or Termination of Approval: If at any point the REC determines that the research is not being conducted in accordance with the approved protocol, or that it is causing unexpected serious harm to participants, it has the authority to suspend or terminate the ethical approval. Suspension is a temporary halt (no new participants, and possibly stopping interventions on current participants except for safety) while an investigation is done. Termination is a permanent stop to the study’s activities under that approval. The REC will promptly notify the investigator (and institutional officials and regulatory bodies) if such a decision is made, providing the reasons. The REC will also advise on steps to protect participants during the withdrawal (for example, ensuring medical follow-up or transition of care out of the research).
- Follow-up Review Documentation: All of the above communications and decisions during the post-approval phase are documented in the REC files. Progress reports and continuing review outcomes are noted in meeting minutes. SAE reports and deviations are logged and filed with any corresponding REC letters or requirements. This ensures that the history of the study’s conduct is recorded.
Through these follow-up procedures, the REC maintains oversight and helps ensure that the study continues to uphold ethical standards throughout its duration.
Record Keeping and Archiving
Accurate records are the backbone of REC operations and are essential for transparency and accountability:
- Protocol Files: For each study that the REC reviews, a dedicated file (physical and/or electronic) is maintained. This file contains the original application, all reviewed documents (protocol, consent forms, tools, etc.), correspondence (letters of approval, emails from the investigator, clarifications, etc.), and any subsequent submissions (amendments, progress reports, SAE reports).
- Minutes: Detailed minutes of each REC meeting are kept, capturing attendance, discussions (without attributing comments to individuals to encourage open dialogue), decisions, and the vote counts or consensus outcome. Once approved at the subsequent meeting, minutes are signed by the Chair (or Secretary) and archived online.
- Membership Records: The REC office keeps records of the committee’s membership, including each member’s CV, appointment letters, training certificates, and signed confidentiality agreements. This demonstrates the committee’s composition meets required qualifications and that members are up-to-date with training.
- SOPs and Guidelines: The current version of the REC’s SOPs is kept on file and accessible to all members. Historical versions are archived as well to track changes over time. Copies of key reference materials (national regulations, international ethical guidelines, etc., including their amendments) are also maintained for reference.
- Correspondence: All incoming and outgoing correspondence related to REC business is filed. This includes communications with investigators (queries and responses), communications with regulatory authorities (like NHRA or ZAMRA), and any other relevant letters or emails.
- Logs and Registers: The REC may maintain summary logs for internal tracking, such as:
- A protocol receipt and processing log (to monitor timelines from submission to decision).
- An approvals register (list of all approved protocols with key details like PI name, title, dates of approval and expiry).
- A training log (dates and topics of training sessions attended by members).
- A finance log (if applicable, recording fees received and any disbursements for REC operations).
- Annual Report: The REC compiles an annual report of its activities, which may include the number of protocols reviewed (approved, rejected, etc.), types of studies, any major issues addressed, changes in membership, and training activities. This report is submitted to LCPTS leadership and the NHRA as required, and may be shared publicly to demonstrate the REC’s contributions to research oversight.
- Archiving Duration: All REC documents are archived for a minimum period of three (3) years after the completion of the research or the last REC action on the protocol. However, in practice, many documents are kept longer (e.g., 5-7 years or per institutional or sponsor requirements). If a study is related to a clinical trial under regulatory oversight, documents might be retained for a longer period in line with those regulations (sometimes 15 years or more, especially if tied to product approvals). The REC will adhere to the longest retention period applicable to ensure compliance.
- Storage and Security: Archived physical files are stored in a secure location (a locked cabinet or room with restricted key access). Electronic archives are kept on secure servers with proper access controls (passwords, limited user access, and routine backups). Sensitive data is protected from unauthorized access, and confidentiality agreements extend to anyone handling these records.
- Confidentiality of Records: All REC records are confidential. Information within protocols (like novel drug information, or personal data about participants) and deliberations are not disclosed outside the REC except to authorized parties (such as regulatory auditors or by legal requirement). When records reach the end of their retention period and are to be disposed, disposal is done securely — paper records are shredded or incinerated, and electronic records are deleted in a manner that prevents recovery.
By diligently maintaining records, the REC ensures that there is an auditable trail of its decisions and actions, which is essential for accountability to participants, researchers, institutional officials, and regulatory bodies.
Ongoing Education and SOP Updates
The LCPTS-REC is committed to continuous improvement and staying current:
- Continual Learning: Members are expected to stay informed about new developments in research ethics. The REC facilitates this by sharing relevant articles, guidance documents, and case studies with members. If there are updates to international guidelines (like a new revision of the Declaration of Helsinki or CIOMS guidelines), these are circulated and discussed.
- Workshops and Conferences: The REC encourages members to attend research ethics trainings, workshops, or conferences when possible. LCPTS may host or sponsor such events, sometimes in collaboration with the NHRA or other institutions. Members who attend external training often share insights with the rest of the committee (for example, by presenting key points learned at a conference).
- SOP Revision: These Standard Operating Procedures are living documents. The REC will review its SOPs at regular intervals (e.g., every two years) or in response to specific needs (like a change in national regulations, or after encountering a novel issue that isn’t well-covered in the current SOP). Revisions are discussed in the committee, and any changes are approved by the appropriate authority (LCPTS Director or Board, and/or NHRA if required). Once adopted, all members are provided with the updated SOPs and trained on any new procedures.
- Quality Assurance: The REC may conduct self-assessments or invite external evaluation to ensure it is functioning optimally. This can include checking if decisions are made within expected time frames, if documentation is complete, or even surveying investigators for feedback on the ethics review process. Findings from such assessments can lead to procedural improvements or additional training.
- Adaptation to Regulatory Changes: If there are changes in the legal or regulatory environment (for example, a new data protection law or updates to the National Health Research Act or its guidelines), the REC will adapt its forms and processes accordingly. Members will be briefed on changes to ensure compliance going forward.
- Collaboration and Networking: LCPTS-REC will maintain communication with other RECs in Zambia and the region to share experiences and harmonize practices. This networking can occur through forums organized by NHRA or research ethics conferences. It helps keep the committee aligned with national standards and informed about how others are handling emerging ethical issues.
- Public Trust: By staying updated and transparent in its processes, the REC aims to maintain public trust. Knowing that the ethics committee is knowledgeable, well-trained, and following the latest guidelines reassures research participants and the community that research overseen by LCPTS-REC is conducted to the highest ethical standards.
SOP Development, Control, and Management
Ownership. The SOPs are owned by the LCPTS-REC and maintained by the Secretary under the oversight of the Chair.
Version Control. Each SOP bears a title, version number, effective date, superseded version, and change log. Only the latest version is “controlled”; older versions are archived.
Change Initiation. Changes may be proposed by any REC member, LCPTS leadership, NHRA requests, or in response to regulatory updates (e.g., CIOMS, 45 CFR 46, ICH-GCP). The Secretary logs proposals in the SOP Change Register.
Review & Approval. Proposed revisions are circulated ≥2-10 working days before a vote. Adoption requires a quorate simple majority. The new version becomes effective upon the Chair’s or director’s signature.
Review Cycle. SOPs undergo biennial review, or sooner if triggered by regulatory changes or audit findings.
Dissemination & Training. The Secretary distributes controlled copies to members within 5 working days of approval and schedules update briefings within 30 days.
References
- Association of Research Ethics Committees (2013). A Framework of Policies and Procedures for University Research Ethics Committees. Association for Research Ethics. [Available online]
- Republic of Zambia (2013). National Health Research Act, 2013. (Act No. 2 of 2013).
- U.S. Department of Health and Human Services (2018). 45 CFR 46: Federal Policy for the Protection of Human Subjects (“Common Rule”).
- World Medical Association (2013). Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects.
